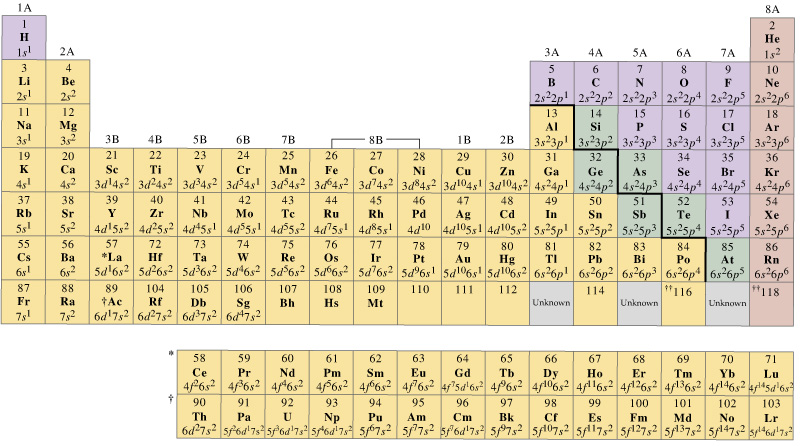
Elements with the same number of valence electrons are grouped together. The number of valence electrons determines an element’s position in the periodic table and its chemical behavior. Valence Electrons Determine the Periodic Table Their interaction determines how atoms combine to form molecules. Valence electrons play a crucial role in the bonding of atoms and the formation of chemical compounds. It determines the chemical properties and reactivity of an element. Valence electron configuration refers to the distribution of electrons in the outermost energy level, also known as the valence shell, of an atom. Whether you are a student, a chemistry enthusiast, or simply curious about the wonders of the atomic world, prepare to be amazed by these engaging facts about valence electron configuration! From the significance of electron shells to the octet rule, electronegativity, and hybridization, we will uncover the secrets behind chemical bonding and its impact on the properties of various elements and compounds. In this article, we will delve into the intriguing world of valence electron configuration and explore 20 fascinating facts about this concept. Understanding the valence electron configuration helps chemists predict the formation of chemical bonds, the stability of compounds, and even the colors and magnetic properties of substances. These electrons, found in the outermost energy level of an atom, determine its reactivity and the way it interacts with other atoms. 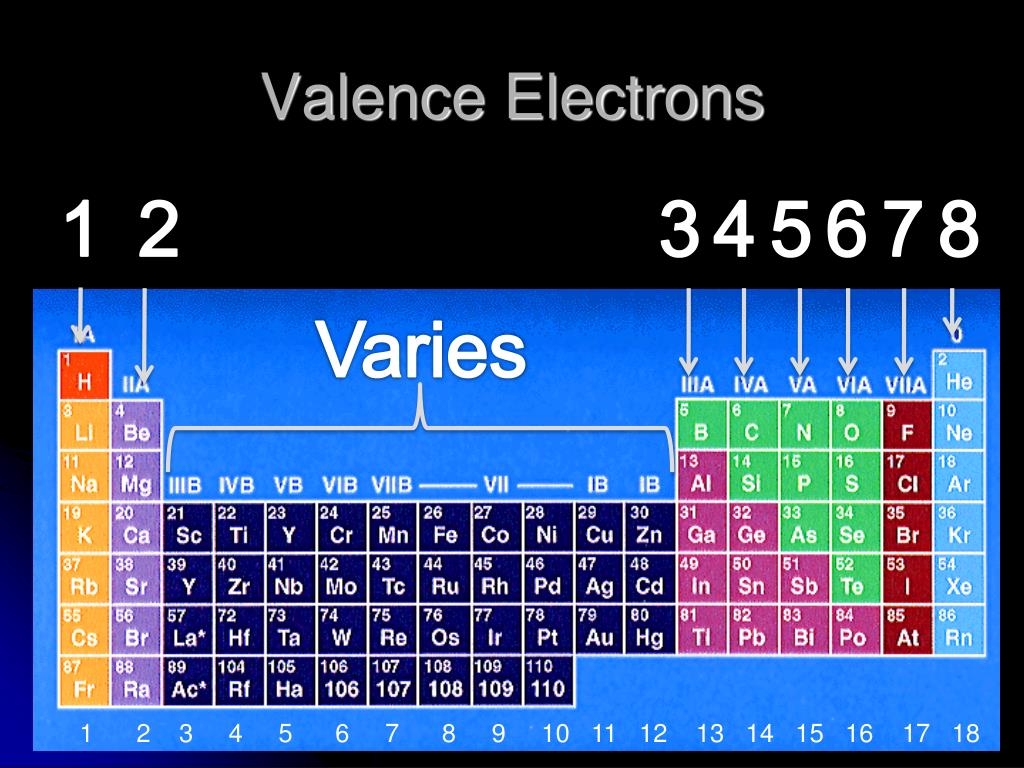
Valence electron configuration is a fundamental concept in chemistry that plays a crucial role in understanding the behavior and properties of elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
